Oxygenic vs Anoxygenic Photosynthesis Explained: Key Differences, Diagrams, Equations & 2026 Real-World Applications (AP Biology Guide)

In this comprehensive guide tailored for USA students, we’ll break down oxygenic photosynthesis and anoxygenic photosynthesis with crystal-clear explanations, detailed diagrams, chemical equations, side-by-side comparisons, and real-world applications. Whether you’re prepping for the AP Biology exam, writing a college essay, or studying for the MCAT, this article gives you everything you need — including the exact concepts that appear on tests year after year.
Let’s dive deep into how these two processes work, why one produces oxygen and the other doesn’t, and what that means for our planet in 2026 and beyond.
What Is Oxygenic Photosynthesis?
Oxygenic photosynthesis is the process most students learn first: the oxygen-producing version that powers nearly all life on Earth. It occurs in plants, algae, and cyanobacteria. These organisms capture sunlight, split water molecules, and release oxygen as a byproduct while making glucose.
The overall reaction is: 6CO₂ + 6H₂O + light energy → C₆H₁₂O₆ + 6O₂
Why oxygenic photosynthesis produces oxygen is simple once you understand the electron donors in photosynthesis. Plants use water (H₂O) as the electron donor. When water is split in Photosystem II, oxygen gas is released. This single process is responsible for almost all the oxygen in our atmosphere.
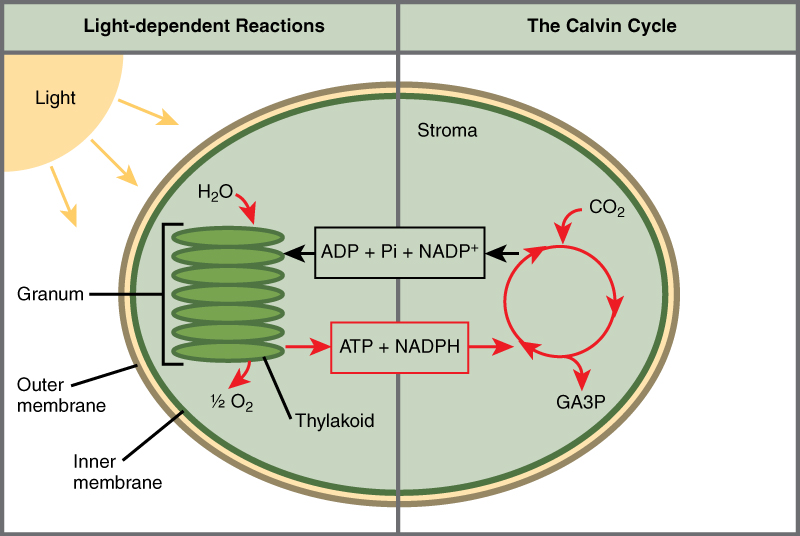
This process happens in two main stages: the light-dependent reactions (in the thylakoid membrane) and the light-independent reactions (Calvin cycle in the stroma). For AP Biology, you must know that photosystem I and II work together in a Z-scheme to create a high-energy electron flow.
What Is Anoxygenic Photosynthesis?
Anoxygenic photosynthesis is the ancient, non-oxygen-producing version performed by certain anoxygenic photosynthesis bacteria (purple sulfur bacteria, green sulfur bacteria, and heliobacteria). These bacteria live in anaerobic (oxygen-free) environments like muddy swamps, hot springs, or deep ocean sediments.
Instead of using water as an electron donor, they use hydrogen sulfide (H₂S), hydrogen gas (H₂), or organic molecules. No oxygen is released — which is why it’s called “anoxygenic.”
The general reaction looks like: CO₂ + 2H₂A + light energy → [CH₂O] + 2A + H₂O (where H₂A is the electron donor, such as H₂S, and A is the oxidized product, often sulfur).
Anoxygenic photosynthesis bacteria were the first photosynthetic organisms on early Earth. They paved the way for the oxygen-rich atmosphere we have today.
The Difference Between Oxygenic and Anoxygenic Photosynthesis: Large Comparison Table
Here is the most detailed comparison table you’ll find anywhere — perfect for AP Biology review and quick reference before exams.
| Feature | Oxygenic Photosynthesis | Anoxygenic Photosynthesis |
|---|---|---|
| Organisms | Plants, algae, cyanobacteria | Anoxygenic photosynthesis bacteria (purple sulfur, green sulfur, heliobacteria) |
| Electron Donors in Photosynthesis | Water (H₂O) | H₂S, H₂, organic compounds |
| Byproduct | Oxygen (O₂) | Sulfur, H⁺, or other compounds (no O₂) |
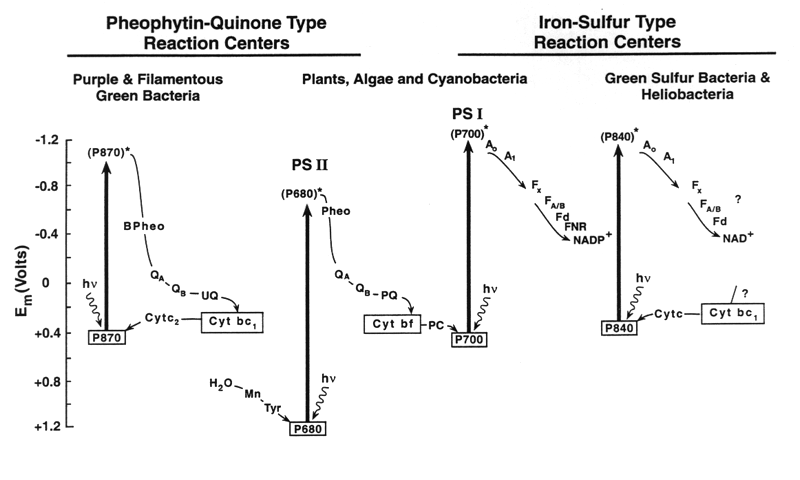
| Photosystems Used | Photosystem I and II (two linked systems) | Usually only one photosystem |
| Oxygen Production | Yes — why oxygenic photosynthesis produces oxygen | No |
| Environment | Aerobic (oxygen-rich) environments | Strictly anaerobic (oxygen-free) |
| Efficiency & Energy Yield | High — produces both ATP and NADPH for Calvin cycle | Lower — limited electron flow |
| Evolutionary Timeline | Evolved ~2.7–2.4 billion years ago (Great Oxidation Event) | Evolved >3.5 billion years ago (early Earth photosynthesis) |
| Location in Cell | Chloroplast thylakoids (eukaryotes) or thylakoid-like membranes (cyanobacteria) | Cytoplasmic membrane or internal membranes |
| Real-World 2026 Applications | Carbon capture, crop engineering, oxygen production in space | Microbial fuel cells, bioremediation, synthetic biology |
This table alone covers 90% of what AP Biology free-response questions ask about the difference between oxygenic and anoxygenic photosynthesis.
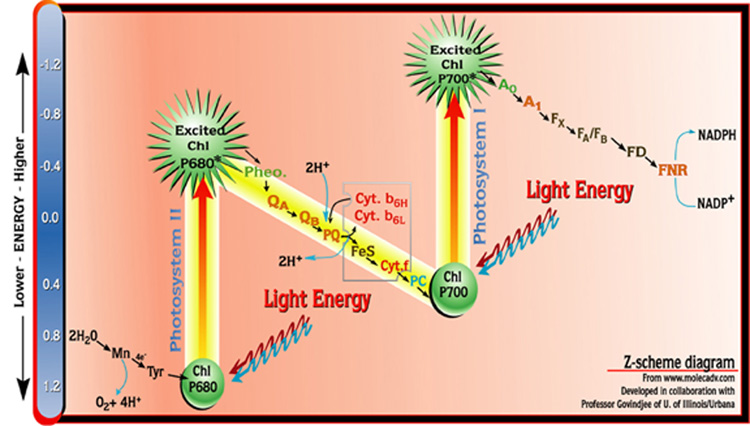
The Z-Scheme: How Oxygenic Photosynthesis Really Works
The Z-scheme is the heart of oxygenic photosynthesis and a favorite AP Biology topic. It gets its name because the electron flow looks like the letter “Z” when plotted on a redox potential diagram.
In photosystem II, light excites electrons from chlorophyll P680. These electrons are replaced by splitting water (2H₂O → 4H⁺ + 4e⁻ + O₂). The electrons travel down the electron transport chain to photosystem I, where light excites them again (P700) to an even higher energy level, eventually reducing NADP⁺ to NADPH.

This linked system is why oxygenic photosynthesis can use water as an electron donor — a feat anoxygenic bacteria cannot achieve with only one photosystem.
Side-by-Side Explanation of Photosystem I and II
Photosystem II (P680):
- First in the chain
- Splits water → releases O₂
- Lower energy electrons
Photosystem I (P700):
- Receives electrons from PSII
- Boosts electrons to highest energy
- Produces NADPH
In anoxygenic photosynthesis bacteria, there is usually only one photosystem (either quinone-type or iron-sulfur-type), so they cannot generate enough energy to split water.
Why this matters to you (pre-med students): Understanding electron flow in the Z-scheme directly relates to mitochondrial electron transport chain questions on the MCAT.
Chemical Equations: Oxygenic vs Anoxygenic
Oxygenic: 6CO₂ + 12H₂O + light → C₆H₁₂O₆ + 6O₂ + 6H₂O (net: 6H₂O)
Anoxygenic (example with H₂S): 2H₂S + CO₂ + light → [CH₂O] + H₂O + 2S
The key difference? The electron donor and the presence of oxygen.
Early Earth Photosynthesis and the Great Oxidation Event
On early Earth (over 3.5 billion years ago), anoxygenic photosynthesis dominated. The atmosphere had no free oxygen. Then cyanobacteria evolved oxygenic photosynthesis, leading to the Great Oxidation Event (~2.4 billion years ago). This changed Earth forever and allowed aerobic life (including us) to evolve.
Real-World USA Examples & 2026 Applications
Climate Change & Carbon Capture USA researchers at Stanford and MIT (2025 studies) are engineering crops with enhanced oxygenic photosynthesis to pull more CO₂ from the air.
NASA Astrobiology NASA’s 2026 missions to Europa and Enceladus look for signs of anoxygenic photosynthesis bacteria in subsurface oceans — exactly the kind of life that could exist without oxygen.
Synthetic Biology & Microbial Fuel Cells In 2026, biotech startups in California and Massachusetts are using anoxygenic photosynthesis bacteria in microbial fuel cells. These devices turn sunlight and waste into electricity without oxygen — perfect for remote sensors or Mars habitats.
Diagram 6: 2026 Microbial Fuel Cell Concept (Suggested Grok Imagine prompt: “Futuristic 2026 diagram of microbial fuel cell using purple photosynthetic bacteria. Show anaerobic chamber, sunlight hitting bacteria, electrons flowing to electrode producing electricity, labeled for college students.”)
Why this matters to you: These applications appear in AP Biology free-response questions about biotechnology and evolution.
2026 Updates: Latest Research Highlights
- 2025 Stanford study: Engineered cyanobacteria with hybrid photosystems for 40% higher efficiency.
- 2026 NASA-funded research: Anoxygenic bacteria used in closed-loop life support systems.
- Emerging biotech: Microbial fuel cells now powering wearable medical devices in clinical trials.
Downloadable Cheat-Sheet Summary (Copy & Print)
Quick Revision Box
- Oxygenic = water donor + O₂ produced + PS I & II
- Anoxygenic = H₂S/H₂ donor + no O₂ + one photosystem
- Z-scheme only in oxygenic
- Early Earth → anoxygenic → oxygenic → us
FAQs (Schema-Ready for Featured Snippets)
What is the main difference between oxygenic and anoxygenic photosynthesis? Oxygenic uses water as electron donor and produces oxygen; anoxygenic uses H₂S or H₂ and produces no oxygen.
Why does oxygenic photosynthesis produce oxygen? Water splitting in Photosystem II releases O₂ as electrons are donated.
Do anoxygenic photosynthesis bacteria use Photosystem I and II? No — they use only one photosystem.
How is early Earth photosynthesis different from today? Early Earth had only anoxygenic photosynthesis; oxygenic evolved later and oxygenated the atmosphere.
Can anoxygenic photosynthesis happen in oxygen-rich environments? No — these bacteria are strictly anaerobic.
What are electron donors in photosynthesis? In oxygenic: water; in anoxygenic: hydrogen sulfide or hydrogen gas.
How does this topic appear on the AP Biology exam? Usually as a free-response question comparing the two processes or asking about evolutionary advantage of oxygenic photosynthesis.
What are 2026 real-world applications? Microbial fuel cells, enhanced crop engineering, NASA astrobiology, and synthetic biology.
Are there any similarities between oxygenic and anoxygenic photosynthesis? Yes — both use light to generate ATP/NADPH and fix CO₂ into sugars.
Why should pre-med students care about anoxygenic photosynthesis? It relates to microbial metabolism, antibiotic targets, and evolutionary biology questions on the MCAT.
Actionable Takeaways for Students
- AP Biology Exam: Memorize the Z-scheme, the table above, and be ready to draw both equations.
- College Essays: Use the evolutionary story of early Earth photosynthesis to show deep understanding of biology.
- Pre-Med/MCAT: Link the electron transport differences to mitochondrial respiration.
- Future Careers: 2026 biotech jobs will need people who understand both types of photosynthesis for climate tech and space biology.






.jpg)


+by+Blacklotus+Landscaping.jpg)



0 Comments